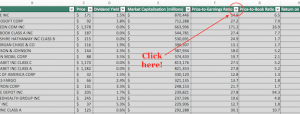
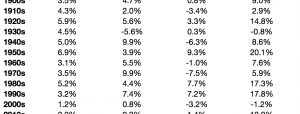
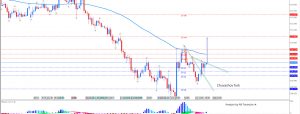
Pfizer (PFE) and Allergan (AGN) announced that the companies have received a request for additional information from the U.S. Federal Trade Commission with respect to their previously announced pending combination.
The request for information from the FTC, often referred to as a “second request,” was fully anticipated as part of the regulatory process under the Hart-Scott-Rodino Antitrust Improvements Act of 1976. The companies continue to work cooperatively and expeditiously with the FTC in connection with its review. Completion of the transaction remains subject to the expiration of the waiting period under the HSR Act and certain other conditions, including governmental and regulatory approvals in certain other jurisdictions, including the EU, the receipt of necessary approvals from both Pfizer and Allergan shareholders, and the completion of Allergan’s pending divestiture of its generics business to Teva Pharmaceuticals (TEVA).
Pfizer and Allergan continue to expect the transaction to close in the second half of 2016.