TM editors’ note: This article discusses a penny stock and/or microcap. Such stocks are easily manipulated; do your own careful due diligence.
On March 15, 2017, BriaCell Therapeutics Corp. (TSXV: BCT) (BCTXF) announced that the U.S. FDA had granted clearance to initiate the Phase 1/2a clinical trial of BriaVax™ in patients with advanced breast cancer. This is an important step for the company and represents many years of hard work by management to get BriaVax back into the clinic, including finalizing the necessary CMC work around a new manufacturing process for the whole-cell vaccine.
I’m expecting the first patients to be dosed shortly. The principal investigator is Dr. Jarrod P. Holmes at St. Joseph Heritage Healthcare in Santa Rosa, California. Dr. Holmes is a Board Certified Oncologist and a leading expert in cancer vaccines. Cancer Insight, LLC, led by Dr. George Peoples, a surgical oncologist and leading expert in cancer vaccines, is managing the clinical study. Biologics Consulting is handling regulatory affairs.
The Phase 1/2a Program
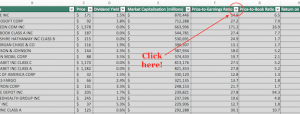
The Phase 1/2a trial (NCT03066947) is expected to enroll up to 24 late-stage cancer patients with recurrent and/or metastatic breast cancer who have failed at least one line of prior therapy (ECOG 0-2). The single-arm, open-label study is designed to determine the optimal dosing regimen and safety profile of the targeted immunotherapy. The primary endpoint is the incidence of treatment-emergent adverse events following multiple injections of BriaVax. The current protocol calls for inoculation of the vaccine at baseline and then every 2 weeks for one month (3 treatments), then monthly for up to one year.
Standard tumor assessments will be performed at baseline and then every 3 months. Important secondary outcome measures include overall survival (OS), objective response rate (ORR), progression free survival (PFS), and durability of tumor response. Other measures including patient quality of life, performance status, weight, and pain will also be measured. After at least 9 subjects have been treated safely, the dose of BriaVax may be escalated or decreased in subsequent patients based on the emerging data. Below is a schematic of the planned protocol:

CEO Steps Up To Fund The Trial
In a move not often seen by micro-cap biopharma companies, BriaCell President and CEO, Dr. William Williams, helped to fund the Phase 1/2a study with this own money. In late February 2017, Dr. Williams entered into a non-brokered private placement of approximately 5.42 million shares of BriaCell common stock at C24¢ per share. Proceeds of approximately C$1.5 million will be used to fund the Phase 1/2a study as well as develop a companion diagnostic platform known as BriaDx. At the time of the closing, Dr. Williams noted that his financial investment, “Reflects my confidence in the long-term value of the company.”
Listen to Dr. Williams interview on UpTickNewswire >> LINK